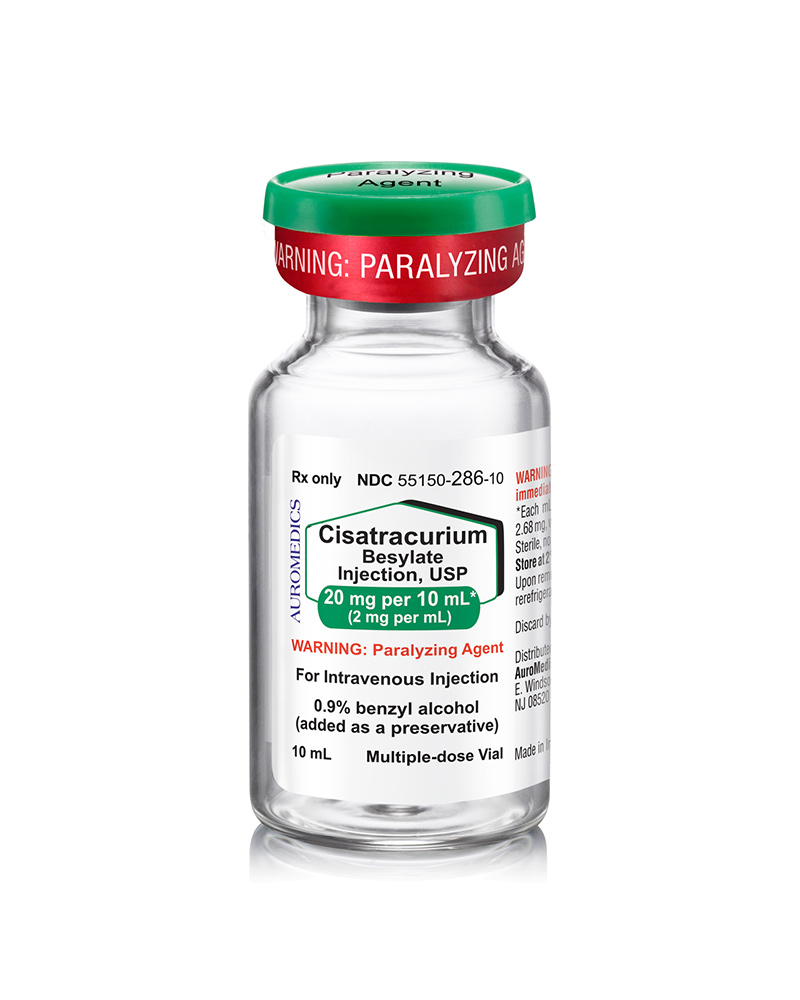
Cisatracurium Besylate Injection
Overview
- Presentation(s):
-
10 mg/5 mL SDV


20 mg/10 mL MDV

- NDC(s):
-
55150-0284-10
55150-0286-10
- Reference Listed Drug:
- Nimbex®
- Therapeutic Class:
- Neuromuscular Blocker
- Physical Form:
- Solution
Product Information
| NDC | Description | Strength | Concentration | Container Size | Fill Volume | Closure |
|---|---|---|---|---|---|---|
| 55150-0284-10 | SD Vial | 10 mg/5 mL SDV | 2 mg/mL | 5 mL | 5 mL | 20 mm |
Wholesaler Item Numbers
ABC
10252328Cardinal
5690102McKesson
2315042M&D
922542Ordering Information
Unit of Sale
10 vialsPack Size
10Case Quantity
20Allergens

| NDC | Description | Strength | Concentration | Container Size | Fill Volume | Closure |
|---|---|---|---|---|---|---|
| 55150-0286-10 | MD Vial | 20 mg/10 mL MDV | 2 mg/mL | 10 mL | 10 mL | 20 mm |
Wholesaler Item Numbers
ABC
10237416Cardinal
5659180McKesson
1555184M&D
906222Ordering Information
Unit of Sale
10 vialsPack Size
10Case Quantity
24Allergens

Additional Information
Bioequivalency Rating
APBar Code Type
LinearControlled Substance Class
N/AStorage Requirements:
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]For Package Inserts, please click on the NDC#
Our products are available from authorized distributors.
For complete ordering information, please contact your local pharmaceutical distributor.